|
4/17/2024 0 Comments Timeline of atomic theory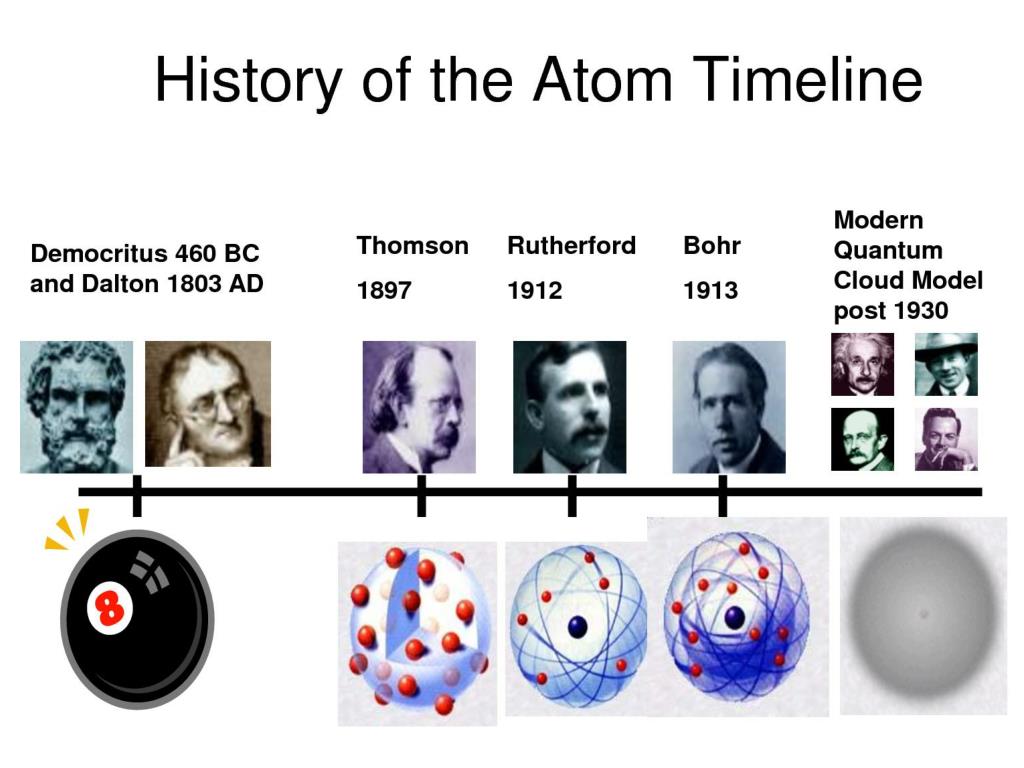 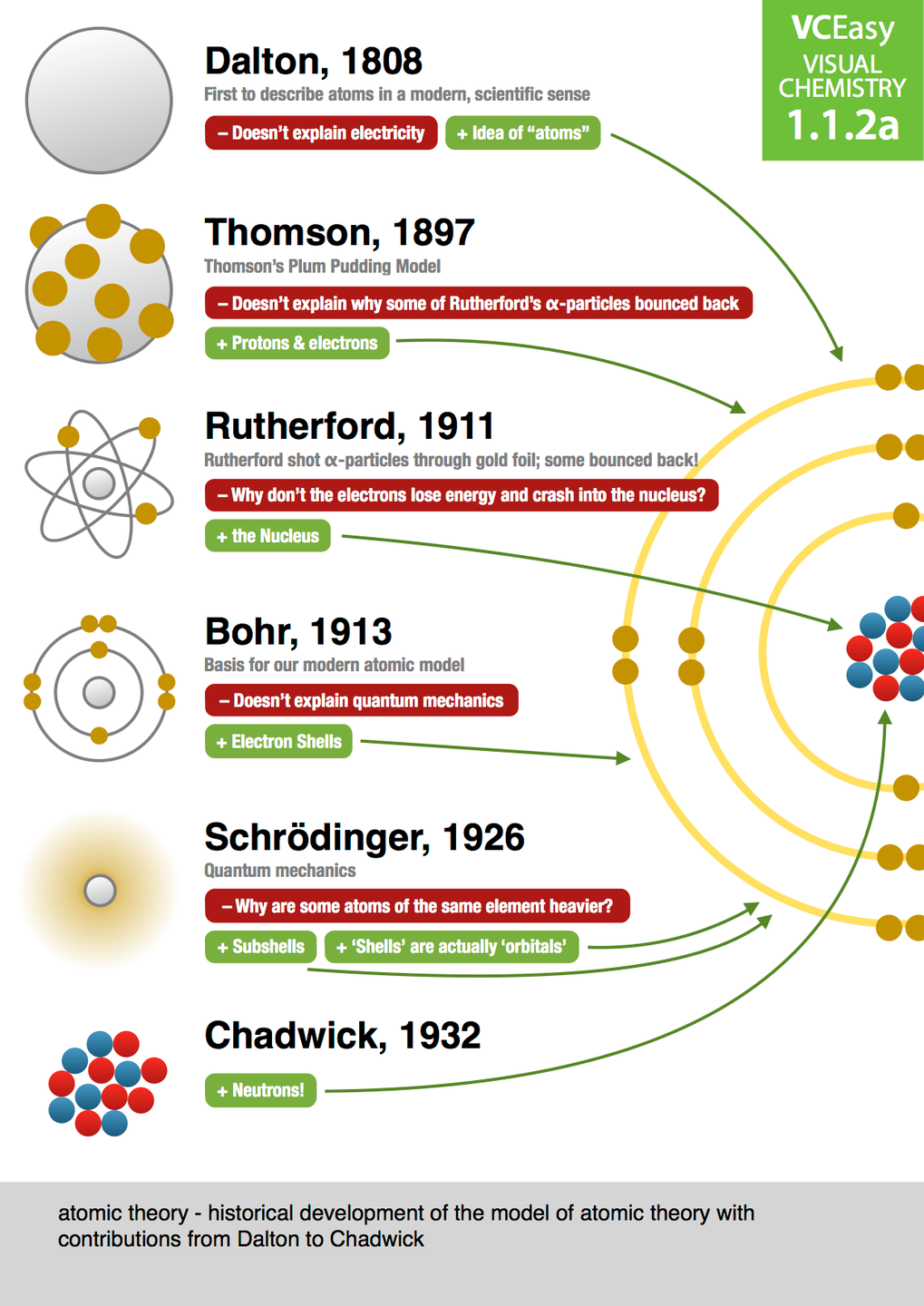 
We've been talking a lot about the fine details ofĬhemistry in recent weeks and we're gonna keep doing that as we move on to nuclear chemistry, and then to the basics But, as has been the case in all science, each scientist built on what As time went on many more were the result of rigorous experimentation. Some models like that of Leucippus were just blind guesses. Hundreds if not thousands of different insights. Theory as we know it today is the product of Then this makes a certain amount of sense if you don't happen to haveĪccess to electron microscopes or cathode ray tubes or the work of generations Clay atoms were softer and attached by ball socket joints that So they thought that iron atoms were hard and stuck together with hooks. Properties of each substance the forms of the atom. Thought that iron was made up of iron particles and clay was made up of clay particles and cheese was made up of cheese particles. The name a tomos, which means uncuttable or indivisible. In half enough times, eventually you'll reach a particle that can't be cut anymore. Special they just thought that if you cut something No one knows how theyĭeveloped this concept but they didn't think the particles were particularly That's when Greek philosopher Leucippus and his pupil Democritus first came up with the idea that matter isĬomposed of tiny particles. And like way more than they thought they knew 2,500 years ago. Know more about atomic theory then the scientists did If you understand enoughĪbout atoms to visualize any of those things then you Moseley discovered that the number of protons in an element determines its atomic number.ġ919 Francis William Aston used a mass spectrograph to identify 212 isotopes.ġ922 Niels Bohr proposed an atomic structure theory that stated the outer orbit of an atom could hold more electrons than the inner orbit.ġ923 Louis de Broglie proposed that electrons have a wave/particle duality.ġ929 Cockcroft / Walton created the first nuclear reaction, producing alpha particlesġ930 Paul Dirac proposed the existence of anti-particles.ġ932 James Chadwick discovered neutrons, particles whose mass was close to that of a proton.ġ938 Lise Meitner, Hahn, Strassman discovered nuclear fission.ġ941-51 Glenn Seaborg discovered eight transuranium elements.ġ942 Enrico Fermi created the first man-made nuclear reactor.- How do you picture an atom in your mind? Like this, or like this, Thomson determined the charge to mass ratio of electrons.ġ898 Rutherford discovered alpha, beta, and gamma rays in radiation.ġ898 Marie Sklodowska Curie discovered radium and polonium and coined the term radioactivity after studying the decay process of uranium and thorium.ġ900 Max Planck proposed the idea of quantization to explain how a hot, glowing object emitted light.ġ900 Frederick Soddy came up with the term “isotope” to explain the unintentional breakdown of radioactive elements.ġ903 Hantaro Nagaoka proposed an atomic model called the Saturnian Model to describe the structure of an atom.ġ904 Richard Abegg found that inert gases have a “stable electron configuration.”ġ906 Hans Geiger invented a device that could detect alpha particles.ġ914 H.G.J. Goldstein discovered canal rays, which have a positive charge equal to an electron.ġ896 Henri Becquerel discovered radiation by studying the effects of x-rays on photographic film.ġ897 J.J. Stoney theorized that electricity was comprised of negative particles he called electrons.ġ879 Sir William Crookes’ experiments with cathode-ray tubes led him to confirm the work of earlier scientists by definitively demonstrating that cathode-rays have a negative charge.ġ886 E. Plucker built one of the first cathode-ray tubes.ġ869 Dmitri Mendeleev created the periodic table.ġ873 James Clerk Maxwell proposed the theory of electromagnetism and made the connection between light and electromagnetic waves.ġ874 G.J. Democritus’ atomic theory posited that all matter is made up small indestructible units he called atoms.ġ704 Isaac Newton theorized a mechanical universe with small, solid masses in motion.ġ803 John Dalton proposed that elements consisted of atoms that were identical and had the same mass and that compounds were atoms from different elements combined together.ġ832 Michael Faraday developed the two laws of electrochemistry.ġ859 J.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
